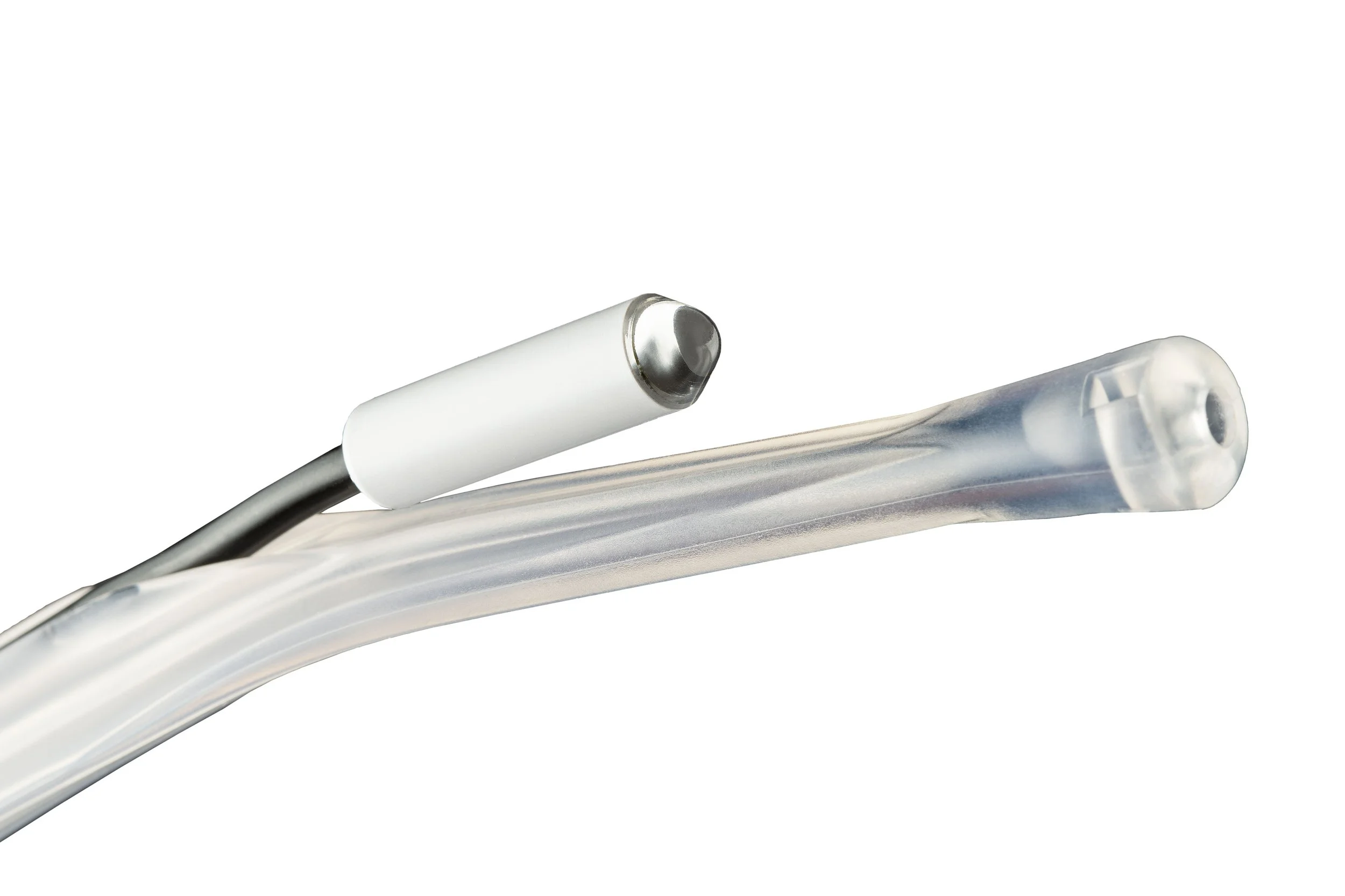
We are a leader in manufacturing handheld LED surgical lights in Phillipsburg, New Jersey. Our lights are able to light up the deepest of cavities, attach to your favorite retractor or tool in the OR and are available on a Yankaeur Suction or Soft Tissue Retractor. Trusted and used by Doctors and Surgeons specializing in Spine, Orthopedics, Obstetrics and Gynecology, Plastics, Colon- Rectal and Emergency Departments for over a decade now.
Shipped Sterilized
No Light Box
No Cables
Individually Packaged
14 KLX at 4”
3 Hour Run Time
IPX5 Resistant
5 Lights per Box
Single Use Disposable
Made in USA
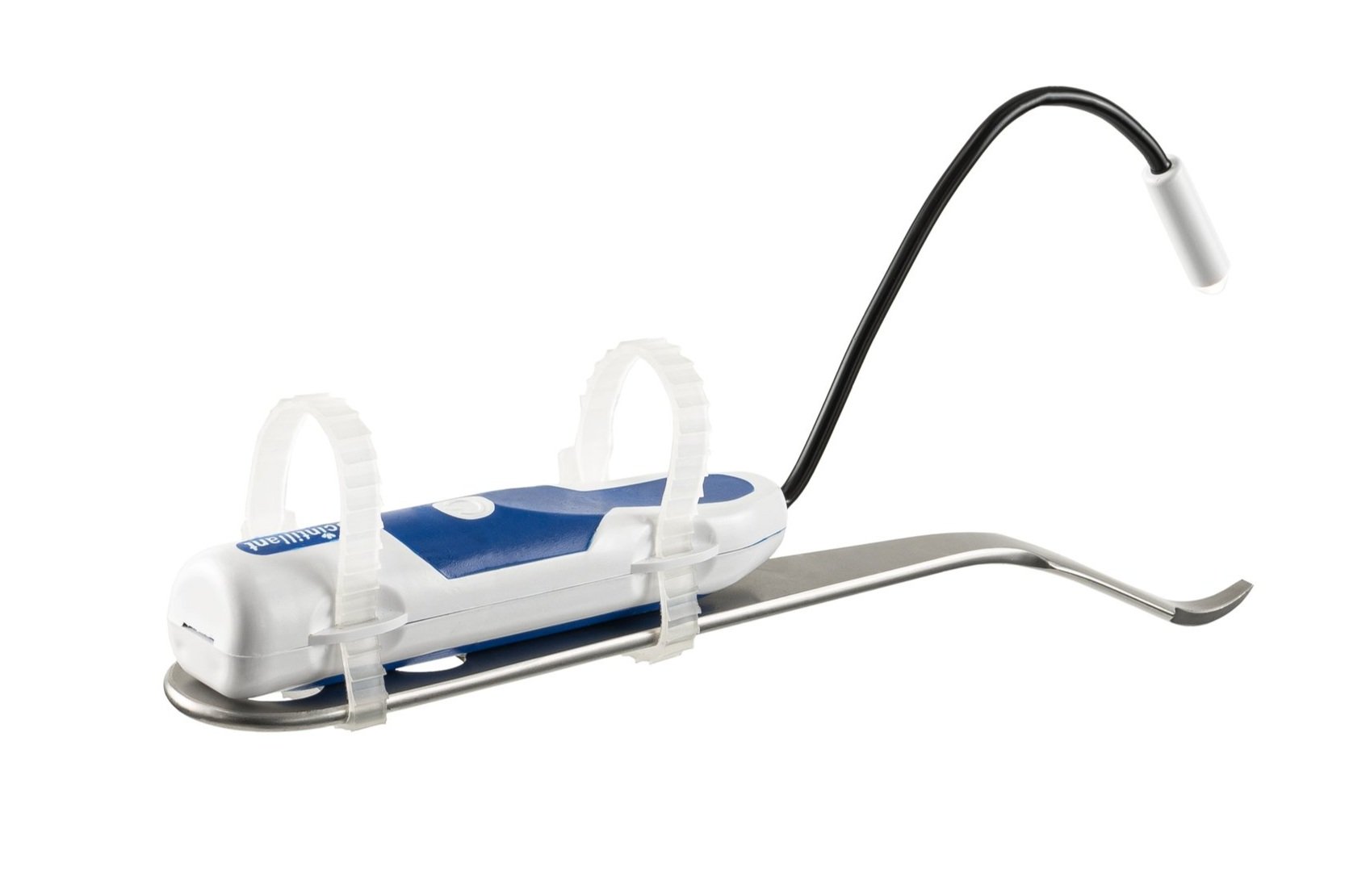
Lighted Suction
The BihlerMed® Surgical Light with Suction (formerly know as the Scintillant Suction) is a handheld surgical light attached to a tonsil-style suction tip, which is designed to combine fluid aspiration with bright LED illumination. The Suction and Light are ready for use directly from the package, immediately enabling users to safely navigate obstructions, to clearly visualize anatomy and to easily aspirate fluids.
Use our Lighted Suction during complex procedures when every second counts, to ensure that you have the visibility you need throughout.
This product is one of the most versatile configurations in the BihlerMed® catalog. Procedures in gynecology and urology, colorectal surgeries, general surgeries, orthopedic surgeries and plastic surgeries have all benefited from the combination of suction and powerful lighting being available in the same instrument.
Shipped Sterilized
No Light Box
No Cables
Individually Packaged
14 KLX at 4”
3 Hour Run Time
IPX5 Resistant
5 Lights per Box
Single Use Disposable
Made in USA
Lighted Retractor
The BihlerMed® Surgical Light with Retractor (formerly knows as Scintillant Soft Tissue Retractor) is a handheld surgical light affixed to a low-profile flat retractor, which combines moderate blunt retraction of tissue with bright LED illumination. The ergonomic design enables users to safely navigate critical obstructions, clearly visualize anatomy and efficiently retract tissue. Shipped sterile, the BihlerMed® Surgical Light with Retractor is ready for use immediately upon removal from its protective packaging.
Use the BihlerMed® Surgical Light with Retractor to ensure that you’re getting the best visualization possible when inspecting the anatomy of your patient. Surgeons have found this device to be highly useful in all gynecological, general, plastic and reconstructive surgical procedures.
Shipped Sterilized
No Light Box
No Cables
Individually Packaged
14 KLX at 4”
3 Hour Run Time
IPX5 Resistant
5 Lights per Box
Single Use Disposable
Made in USA
Zip-Strap
The BihlerMed® Surgical Light with Zip-Strap (formally know as Scintillant Zip-Strap) is a handheld LED surgical light featuring rubber straps for simple attachment to surgical instruments. The non-tethered configuration of the Surgical Light with Zip-Strap enables users to enhance their operating room instruments, to clearly illuminate the surgical field and to safely navigate critical obstructions during surgical procedures. The innovative design of the BihlerMed® Surgical Light with Zip-Strap provides a wide range of opportunities to combine its bright white illumination with standard operating room instruments.
The Zip-Strap will effectively supply your surgeons with light bright illumination, even in the most complex of surgical sites, such as affixing to a retractor for delicate spinal procedure, or total hip revision: for example, affixing the BihlerMed® Surgical Light with Zip-Strap’s to a retractor to light up the inside of a hip cavity. Whatever instruments you must use to carry out your procedures, BihlerMed® Lights will ensures that you will use those tools in a site that that is brimming with vivid illumination.
Dual Tip Surgical Lights
BihlerMed® offers the Dual Tip Surgical Lights in two distinct models: the Straight Tip and the Bent Tip. Each model boasts two separate 20-inch long wands, each fitted with an LED at its tip. The dimensions of the light diameter and wand length make them particularly well-suited for a variety of applications, including the illumination of spinal retractor blades.
Thanks to their non-tethered design, these lights empower users to effortlessly illuminate the surgical field and navigate around critical obstructions during procedures.
In complex surgeries, the BihlerMed® Dual Tip Surgical Lights ensure continuous illumination for your surgeons. They have proven particularly effective in providing deep cavity illumination during delicate spinal procedures, showcasing a unique and invaluable capability. With BihlerMed®, you can trust that your surgical space will remain brilliantly lit, providing your team with the tools they need for success.
Shipped Sterilized
No Light Box
No Cables
Individually Packaged
14 KLX at 4”
3 Hour Run Time
IPX5 Resistant
5 Lights per Box
Single Use Disposable
Made in USA
Medical Device Manufacturing
BihlerMed® is an innovative leader in LED surgical light manufacturing, and in custom medical device manufacturing. As a member of the Bihler® Group of Companies, we can deliver technological solutions that draw on the vast repository of Bihler® knowledge and experience.
We provide services such as: research and
development, design and engineering, and testing, clean room packaging, sterilization coordination, in addition to manufacturing. Our trusted performance will enable you to meet the critical needs of the medical industry, and a wide array of applications.
Get started with BihlerMed, today.

Stay in touch with BihlerMed
Events
ACOG 2024 Meeting
May 17-19th, 2024 | San Francisco, CA
Moscone Center
747 Howard St. San Francisco, CA.
The must-see orthopedics event of 2024 is just around the corner. Please stop by and visit our booth #1040, see you there!